Aggressive brain tumor mapped in genetic, molecular detail
Glioblastoma study could lead to new treatments, including better immunotherapies
 Albert H. Kim
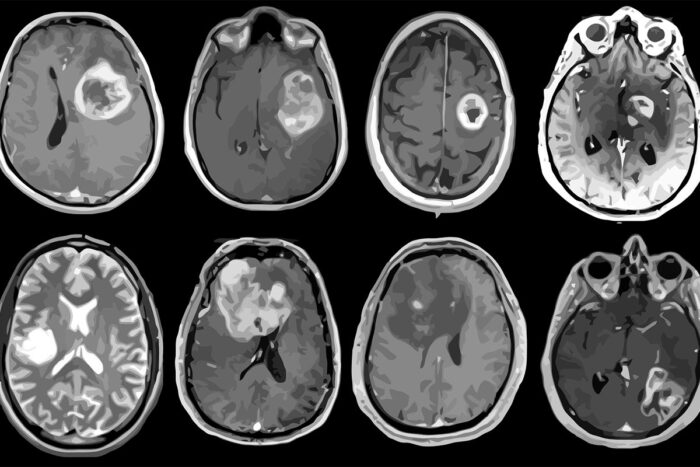
Albert H. KimPictured are MRI scans of eight patients with glioblastoma, an aggressive brain tumor. A new study led by Washington University School of Medicine in St. Louis has mapped out detailed molecular and genetic schematics of these tumors, opening the door to potential improved therapies.
Glioblastoma is among the most aggressive and devastating of cancers. While rare compared with other cancers, it’s the most common type of brain cancer. Even with intensive therapy, relatively few patients survive longer than two years after diagnosis, and fewer than 10% of patients survive beyond five years. Despite extensive studies focused on genomic features of glioblastoma, relatively little progress has been made in improving treatment for patients with this deadly disease.
Now, a new study led by Washington University School of Medicine in St. Louis, Pacific Northwest National Laboratory, Case Western Reserve University and the National Cancer Institute (NCI) of the National Institutes of Health (NIH) has revealed a detailed map of the genes, proteins, infiltrating cells and signaling pathways that play key roles in driving glioblastoma. The study, of 99 tumors from patients, is the largest and most detailed schematic of this deadly brain tumor.
The research, published Feb. 11 in the journal Cancer Cell, is part of the NCI’s Clinical Proteomic Tumor Analysis Consortium (CPTAC).
“To improve therapies for this deadly cancer, understanding the tumor cells themselves is important but not enough,” said senior author Li Ding, PhD, a professor of medicine and of genetics and director of computational biology in the Division of Oncology at Washington University. “We also must understand the tumor cells’ interactions with the surrounding environment, including immune cells and the connective tissues and blood vessels. In our study, we performed high-resolution and high-depth analyses on 99 glioblastoma tumors. Harnessing new technologies, including proteomics, metabolomics and single cell sequencing, this study is an extremely deep dive into glioblastoma tumor biology, revealing new possibilities for therapy.”
The study identified new activated proteins — particularly PTPN11 and PLCG1 — that serve as signaling hubs driving tumor growth in some patients; revealed gene expression patterns involved in a process called epithelial-to-mesenchymal transition that is common in tumor formation; identified four different categories by which to classify glioblastoma, based on the number and types of immune cells present in the tumors; and determined how an understudied protein modification, acetylation, may explain some functional differences between glioblastoma subtypes.
“Acetylation changes a protein’s shape and often results in opening up DNA-protein complexes to facilitate gene expression. By adding protein acetylation to our study, we were able to complete the loop from proteins to genes and gene expression, shedding light on important regulatory changes in glioblastoma,” said co-senior author Karin Rodland, PhD, chief scientist for biomedical research at Pacific Northwest National Laboratory.
“The most immediate implications for these findings are better design of clinical trials,” said co-author Milan G. Chheda, MD, an assistant professor of medicine who treats patients at Siteman Cancer Center at Barnes-Jewish Hospital and Washington University School of Medicine. “For most clinical trials, we take all comers and give them the same treatment. We are not designing trials in the most precise way because we have not fully understood the molecular differences between each patient’s tumor. This leads us to call a treatment a failure when in fact it may be helping specific people.”
The new study demonstrates that individual tumors are likely to respond differently to targeted therapies. For example, the immune landscape of these tumors varied widely, fitting into four separate categories. Type 1 tumors contain high numbers of immune cells called macrophages and a few T cells. Type 2 tumors have a moderate number of macrophages. Type 3 tumors include high numbers of T cells and a few macrophages. And type 4 tumors are what Ding calls an immune desert, with few or no immune cells of any type. So, an immunotherapy that targets macrophages, for example, might work well in patients with type 1 tumors but not at all in patients with type 4. Still, a clinical trial that lumps all patients together may not show such a drug works at all, when averaged across all patients.
“What’s especially appealing about this study is the clustering of glioblastoma into four groups based on immune subtypes that emerged by combining proteomic and genomic comprehensive analysis,” said Henry Rodriguez, director of the Office of Cancer Clinical Proteomics Research at the NCI. “This may open the door for effective responses to immune therapies.”
Added co-author Albert H. Kim, MD, PhD, a professor of neurological surgery at Washington University and director of the Brain Tumor Center at Siteman: “Immunotherapy clinical trials in glioblastoma have been negative so far. And the fact that there are four different immune subgroups may be one of the reasons behind that. We can’t treat all glioblastoma tumors as one disease.”
A group led by co-senior author Tao Liu, PhD, of Pacific Northwest National Laboratory, measured all of the proteins in the tumor samples as well as two specific modifications, called phosphorylation and acetylation, that affect biological functions such as cell signaling. Adding these data into the genomic analysis of the tumors revealed a small subset of glioblastomas that did not fit neatly into any of the typical genomic subtypes. These mixed-subtype tumors were associated with a poor clinical outcome, providing the researchers with clues to the factors affecting the aggressiveness of a tumor that were not evident from genetic information alone.
“These patterns provide additional information for researchers to understand how the glioblastoma subtypes they identified may vary in biological function,” Liu said. “This multifaceted analysis provides an unprecedented level of detail, which is starting to connect the missing dots in glioblastoma.”
Added Chheda, “This paper is an example of the advances that can be made when there is deep collaboration between many experts across the country who the National Cancer Institute has the ability to bring together.”
The researchers are conducting further studies to identify the best drugs to investigate in glioblastoma patients, depending on where their diseases fall on the new tumor map.
The study’s co-first authors are Liang-Bo Wang, a doctoral student in Ding’s lab; Alla Karpova, a doctoral student in Ding’s lab; Song Cao, PhD, an instructor in medicine; and Yize Li, a doctoral student in Ding’s lab, all of Washington University School of Medicine; and Marina A. Gritsenko, and Jennifer E. Kyle, PhD, both of Pacific Northwest National Laboratory.