Bacteria, viruses in gut linked to severity of HIV infection
Intestinal microbes may speed progression from HIV to AIDS
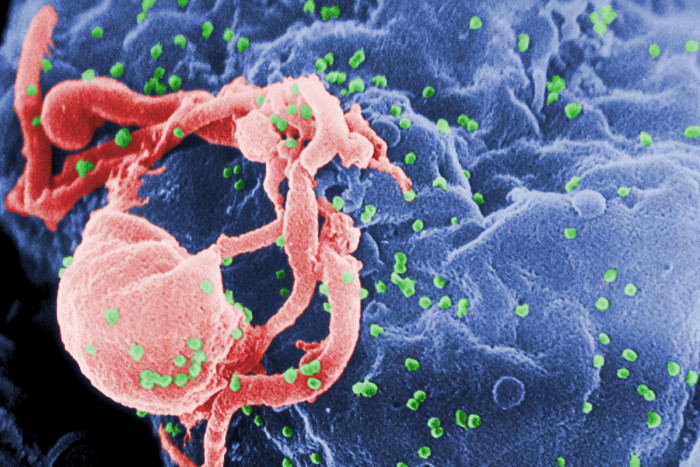 C. Goldsmith
C. GoldsmithA picture taken through an electron microscope shows the HIV virus in an immune cell. Two new studies led by researchers at Washington University School of Medicine in St. Louis point to intestinal bacteria and viruses as possible sources of inflammation and disease in those with HIV infections.
The advent of antiretroviral therapy — a combination of medications used to slow the progression of HIV — has allowed many people infected with the virus to live long, productive lives. But the therapy doesn’t cure them, and those who take the drugs have an elevated risk for cardiovascular disease, cancer, kidney and liver disease, and other disorders seen in HIV patients.
HIV infection also can lead to diseases affecting the intestines, with increased gastrointestinal (GI) inflammation, diarrhea and problems with nutrient absorption. The role of gut microbes in such issues is not completely understood, but now, in two studies led by researchers at Washington University School of Medicine in St. Louis, scientists have identified intestinal bacteria and viruses as possible sources of such inflammation and disease.
Identifying the source could open the door to strategies to limit damage in the GI tract and reduce inflammation and related problems that affect patients with HIV. The two studies — one in people and the other in primates — are published March 9 in the journal Cell Host & Microbe.
“People with advanced HIV have intestinal disease and significant systemic inflammation that may cause progressive defects in immunity and speed the progression from HIV infection to AIDS,” said senior investigator Herbert W. “Skip” Virgin IV, MD, PhD. “We think changes in the virome and bacterial microbiome damage gut epithelial cells and allow bacteria and viruses to leak into surrounding tissues and blood, contributing to inflammation.”
Inflammation is believed to play a role in a range of disorders, including cardiovascular disease, abnormal blood lipids, diabetes and other insulin-related abnormalities.
One of the two studies monitored infected primates, and the other followed HIV-positive people in the African country of Uganda. Both showed that changes in the viral and bacterial makeup of the GI tract were linked to severe immunodeficiency due to progression of HIV in human patients and SIV (Simian immunodeficiency virus) in primates.
The researchers studied 36 rhesus monkeys, some of which were infected with SIV. They also followed 82 HIV-infected people in Uganda, 40 of whom took antiretroviral therapy and 42 of whom did not.
In the people and primates, AIDS and resulting immunodeficiency were linked to a greater number of viruses in the gut, compared with the number of gut viruses in people and primates who had a normal immune response. Those with severely weakened immune systems also had more bacterial pathogens in the gut. In the study of SIV in monkeys, the researchers found that vaccination against SIV prevented the development of those abnormalities.
The severity of immunodeficiency is determined in humans by measuring counts of immune cells called CD4 cells. A count of 200 or lower means that person’s immune system is deficient. Significantly, HIV infection alone, in the absence of immunodeficiency, had a minimal effect on the number of viral and bacterial pathogens in the gut.

Virgin, the Edward Mallinckrodt Professor and head of the Department of Pathology and Immunology, said controlling the proliferation of damaging viruses and bacteria in the gut might reduce damage in the GI tract and, as a result, limit problems that affect patients with chronic HIV infection.
“In these studies, we’ve learned more about which viruses emerge and when they emerge following infection with SIV and HIV,” said Scott Handley, PhD, assistant professor of pathology and immunology and the first author on the study focused on infections in primates. “We’ve also identified potentially disease-causing bacteria in the gut that appear only in severely immunocompromised people and animals.”
Specifically, the researchers found AIDS and resulting immunodeficiency are associated with the expansion of adenoviruses and enterobacteriacae. Both are enteropathogens, meaning they have the potential to cause disease.
“It eventually may be possible, perhaps by treating patients with a specialized mixture of probiotics, to help restore gut health and prevent the damage to cells in the gut that contributes to inflammation and to an HIV infection becoming AIDS,” said co-investigator Guoyan Zhao, PhD, an assistant professor of pathology and immunology.
“There are multiple, novel viruses in the intestinal ecosystems of those with SIV and HIV infection,” Virgin added. “Some of those viruses, particularly the adenoviruses that can damage intestinal cells, also may have the capacity to act as vectors for vaccine delivery. Some of our collaborators are investigating that strategy and are working to develop a vaccine.”