Chemical compound clears cellular waste, protects neurons in model of frontotemporal dementia
Restoring autophagy is promising strategy for range of neurodegenerative diseases
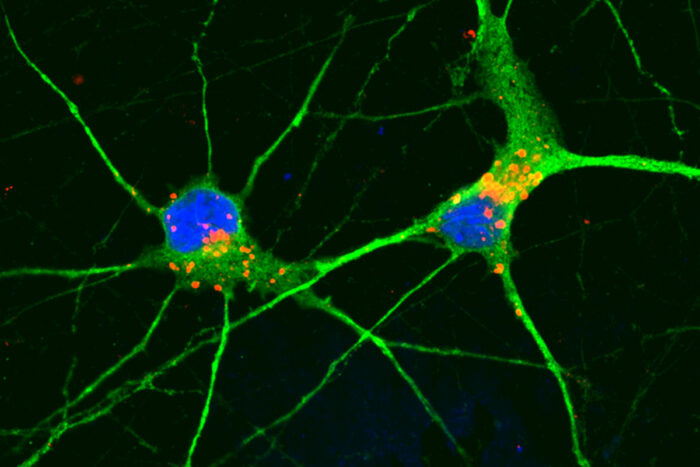 Farzané Mirfakhar
Farzané MirfakharResearchers at WashU Medicine have shown that a novel compound they developed can clear a harmful protein from human neurons modeling frontotemporal dementia (shown) and prevent those neurons from dying.
New research from Washington University School of Medicine in St. Louis adds to growing evidence that helping brain cells break down and eliminate their own cellular waste is a promising treatment strategy for a variety of neurodegenerative diseases. In lab experiments, the researchers found that exposure to a novel compound can clear a harmful protein from human neurons modeling frontotemporal dementia — a devastating and ultimately fatal condition — and prevent those neurons from dying.
The study is published March 31 in the journal Nature Communications.
According to the researchers, the study’s results provide further evidence that enhancing autophagy, a key cellular process involved in breaking down and recycling cellular waste, could help treat neurodegenerative diseases. Autophagy is known to decline with age, so strategies to restore it could help address multiple age-related diseases.
The researchers, led by Celeste Karch, PhD, the Barbara Burton and Reuben M. Morriss III Professor in the WashU Medicine Department of Psychiatry, studied a specific mutation in a brain protein called tau that causes the protein to become misfolded and alter its normal function. In general, when tau proteins become misfolded, they build up inside neurons and contribute to various forms of dementia, including Alzheimer’s dementia and frontotemporal dementia, a neurodegenerative disease similar to Alzheimer’s that often strikes earlier — in middle age — and typically involves significant changes in personality and behavior that precede cognitive decline.
“We found that this tau mutation can clog the cell’s normal cellular clean-up system and interfere with how cells in the brain clean up misfolded proteins,” Karch said. “One compound in particular had an impressive effect in making the cells look almost normal in their clearance of misfolded proteins. In the future, we can envision new therapies for neurodegenerative diseases that may be similar to what we have now for cancer: multi-pronged treatments that combine several drugs attacking different aspects of the disease simultaneously.”
For example, in Alzheimer’s disease, a therapy designed to enhance autophagy could help eliminate tau and, in theory, be combined with FDA-approved antibody therapies that reduce amyloid beta, another damaging Alzheimer’s protein that builds up in neurons and contributes to dementia. More broadly, an autophagy-based approach that clears misfolded proteins could be combined with drugs such as antisense oligonucleotides that reduce production of those same proteins, thus creating a complementary, two-pronged strategy.
The tau mutation analyzed in the current study was first identified in 1998 by researchers at the WashU Medicine Knight Alzheimer Disease Research Center. In this new study, the researchers studied neurons that had been reprogrammed from skin cells sampled from patients with frontotemporal dementia who carried the tau mutation. In the neurons, the mutated tau proteins caused waste-recycling centers called lysosomes, which are involved in autophagy, to become dysfunctional, allowing cellular waste to accumulate in the lysosomes, which may contribute to neuronal death. The researchers found that enhancing autophagy with an analog of the chemical compound G2 improved clearance of the garbage, reduced tau levels in the lysosomes and prevented cellular toxicity and death.
G2 was discovered in 2019 in the labs of WashU Medicine co-authors David H. Perlmutter, MD, executive vice chancellor for medical affairs, the George and Carol Bauer Dean of WashU Medicine, and the Spencer T. and Ann W. Olin Distinguished Professor; Gary A. Silverman, MD, PhD, the Harriet B. Spoehrer Professor and head of the Department of Pediatrics; and Stephen C. Pak, PhD, a professor of pediatrics in the Division of Newborn Medicine. G2 was identified via screening experiments seeking drugs that could reduce the accumulation of an aggregation-prone protein in a C. elegans model of alpha-1-antitrypsin deficiency, which can cause severe liver disease. The compound was later shown to boost autophagy function in mammalian cell model systems.
WashU Medicine researchers also have shown that G2 can protect brain cells from death in cells modeling Huntington’s disease, a fatal inherited neurodegenerative disease caused by a genetic mutation present at birth. In the cellular model of Huntington’s disease, the compound prevented the buildup of a harmful RNA molecule, according to work led by WashU Medicine’s Andrew S. Yoo, PhD, the Philip and Sima K. Needleman Distinguished Professor in the Department of Developmental Biology.
“It’s exciting to see that this compound has protective effects in the context of multiple neurodegenerative diseases,” Karch said. “G2 seems to have similar protective effects even when different dysfunctional proteins are building up in different types of cells.”
With evidence that G2 helps cells clear harmful molecules across multiple cell types implicated in different diseases, it and similar compounds are attractive candidates for preventing cell death, including neurodegeneration, in diseases driven by the toxic buildup of misfolded proteins or other damaging molecules.
In the future, Karch and her colleagues plan to continue evaluating the effectiveness of G2 in clearing misfolded proteins caused by a variety of tau mutations and in multiple types of brain cells.







