Enhanced brain cells clear away dementia-related proteins
New cellular immunotherapy approach for Alzheimer’s disease shows promise in mice
 Yun Chen
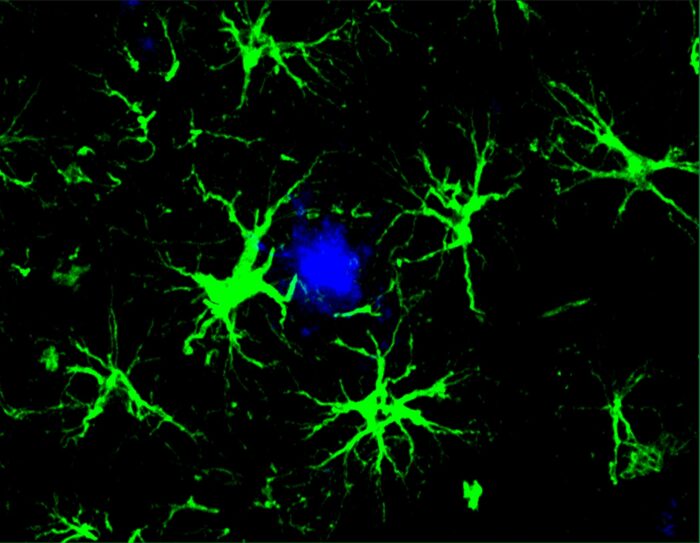
Yun ChenWashU Medicine researchers designed a cellular immunotherapy that turns astrocytes (green), a type of cell in the brain, into super cleaners that sweep away Alzheimer’s-related proteins. With this new feature, the cells successfully reduced the amount of harmful amyloid beta plaques (blue) in the brains of mice.
The new generation of Alzheimer’s disease drugs — the first proven to change the course of the disease — typically extend independent living for patients by 10 months. Called monoclonal antibodies, they reduce the accumulations of a harmful protein, amyloid, in the brain and require high-dose, once- or twice-monthly infusions of the medication.
Now, to reduce the frequency of treatment and potentially improve the efficacy of an anti-amyloid therapy, researchers at Washington University School of Medicine in St. Louis have engineered a new cellular immunotherapy that requires just a single injection to prevent amyloid plaques from developing when given before plaques start to form in mice. Furthermore, a single treatment in mice that had already developed plaques cut the amount of amyloid plaques in half.
The study was published March 5 in Science.
Like CAR-T cell therapies used for cancer treatment, in which T cells of the immune system are genetically modified to attack cancer cells, this new approach equips cells — in this case, brain cells called astrocytes — with a CAR homing device to grab onto a target for destruction. These new CAR-astrocyte cells have features that transform them into super cleaners that remove damaging proteins from the brain that play a role in cognitive decline.
“This study marks the first successful attempt at engineering astrocytes to specifically target and remove amyloid beta plaques in the brains of mice with Alzheimer’s disease,” said the study’s senior author, Marco Colonna, MD, the Robert Rock Belliveau, MD, Professor of Pathology at WashU Medicine. “Although more work needs to be done to optimize the approach and address potential side effects, these results open up an exciting new opportunity to develop CAR-astrocytes into an immunotherapy for neurodegenerative diseases and even brain tumors.”
Removing brain waste
Alzheimer’s disease starts with a sticky protein called amyloid beta that builds up into plaques in the brain, setting off a chain of events that results in brain atrophy and cognitive decline. Microglia, immune cells that reside in the brain, are responsible for removing brain waste but can become dysfunctional when overwhelmed in the context of neurodegenerative disease.
To reduce the cleaning burden on microglia, first author Yun Chen, PhD, then a graduate student in the labs of Colonna and David M. Holtzman, MD, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology at WashU Medicine, transformed astrocytes, the most abundant cell type in the brain, into amyloid-cleaning machines. He custom-designed and delivered a gene to astrocytes that codes for the chimeric antigen receptor (CAR) via a harmless virus injected into mice. The CAR, now present on the surface of astrocytes, enabled the cells to capture and engulf amyloid beta proteins. With their newly acquired ability, the astrocytes — generally responsible for keeping the brain tidy — concentrated their efforts on only cleaning amyloid beta plaques in mice prone to its buildup.
Mice carrying genetic mutations that increase people’s risk of developing Alzheimer’s disease develop amyloid beta plaques that saturate the brain by six months of age. Chen, now a postdoctoral researcher in the Holtzman lab, injected two groups of mice with the virus carrying the CAR-expressing gene: young mice before they developed plaques and older mice with brains saturated with plaques. Then he waited three months.
As the younger mice aged, the CAR-astrocytes prevented amyloid beta plaque development. At nearly six months of age, when untreated mice normally have brains saturated with harmful plaques, brains of treated mice were plaque-free. Meanwhile, older mice with plaque-saturated brains at the time of treatment saw a 50% reduction in the amount of amyloid beta plaques compared to mice receiving an injection of a virus lacking the CAR gene.
The researchers have filed a patent, with help from the Office of Technology Management at WashU, related to the approach used to engineer CAR-astrocytes.
“Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease,” said Holtzman, who is a co-author on the paper. “But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice.”
In future studies, the authors aim to continue improving their CAR-astrocyte immunotherapy by fine-tuning its design to better target harmful proteins, while ensuring no harmful effects on normal brain cell functions. Additionally, by adjusting the CAR homing device to recognize specific markers on brain tumors, they could potentially switch astrocytes’ function from cleaning up debris to directly killing tumor cells. Such an approach could offer a promising new way to treat brain tumors and other central nervous system diseases.