Experimental drug supercharges medicine that reverses opioid overdose
Adding newly ID’d compound makes naloxone more potent, longer lasting, mouse study shows
 GETTY IMAGES
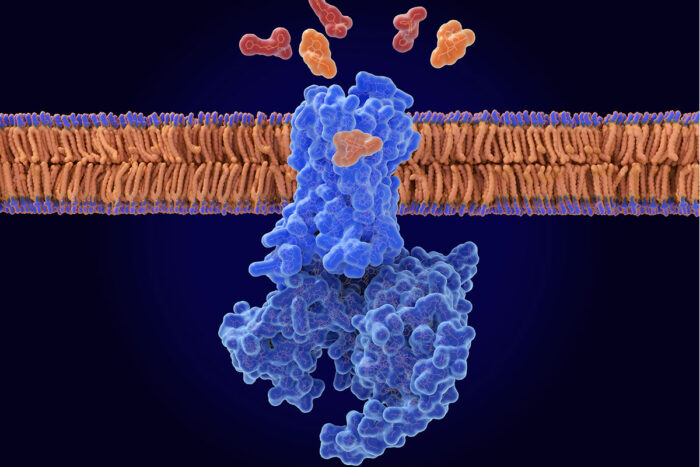
GETTY IMAGESThis illustration shows naloxone (orange) displacing opioids (red) from the opioid receptor (blue) in a nerve cell membrane. Researchers at Washington University School of Medicine in St. Louis, with collaborators at the University of Florida and Stanford University, identified a compound that, in mice, makes naloxone much more effective at counteracting a drug overdose.
The ongoing opioid epidemic in the U.S. kills tens of thousands of people every year. Naloxone, sold under the brand name Narcan, has saved countless lives by reversing opioid overdoses. But new and more powerful opioids keep appearing, and first responders are finding it increasingly difficult to revive people who overdose.
Now, researchers have found an approach that could extend naloxone’s lifesaving power, even in the face of ever-more-dangerous opioids. A team of researchers from Washington University School of Medicine in St. Louis, Stanford University and the University of Florida have identified potential drugs that make naloxone more potent and longer lasting, capable of reversing the effects of opioids in mice at low doses without worsening withdrawal symptoms. The study is published July 3 in Nature.
“Naloxone is a lifesaver, but it’s not a miracle drug; it has limitations,” said co-senior author Susruta Majumdar, PhD, a professor of anesthesiology at Washington University. “Many people who overdose on opioids need more than one dose of naloxone before they are out of danger. This study is a proof of concept that we can make naloxone work better — last longer and be more potent — by giving it in combination with a molecule that influences the responses of the opioid receptor.”
Opioids such as oxycodone and fentanyl work by slipping inside a pocket on the opioid receptor, which is found primarily on neurons in the brain. The presence of opioids activates the receptor, setting off a cascade of molecular events that temporarily alters how the brain functions: reducing the perception of pain, inducing a sense of euphoria and slowing down breathing. It is this suppression of breathing that makes opioids so deadly.
The molecular compound described in the paper is a so-called negative allosteric modulator (NAM) of the opioid receptor. Allosteric modulators are a hot area of research in pharmacology, because they offer a way to influence how the body responds to drugs by fine-tuning the activity of drug receptors rather than the drugs themselves. Co-author Vipin Rangari, PhD, a postdoctoral fellow in the Majumdar lab, did the experiments to chemically characterize the compound.
Naloxone is an opioid, but unlike other opioids, its presence in the binding pocket doesn’t activate the receptor. This unique feature gives naloxone the power to reverse overdoses by displacing problematic opioids from the pocket, thereby deactivating the opioid receptor. The problem is that naloxone wears off before other opioids do. For example, naloxone works for about two hours, while fentanyl can stay in the bloodstream for eight hours. Once naloxone falls out of the binding pocket, any fentanyl molecules that are still circulating can re-attach to and re-activate the receptor, causing the overdose symptoms to return.
The research team — led by co-senior authors Majumdar; Brian K. Kobilka, PhD, a professor of molecular and cellular physiology at Stanford University; and Jay P. McLaughlin, PhD, a professor of pharmacodynamics at the University of Florida — set out to find NAMs that strengthen naloxone by helping it stay in the binding pocket longer and suppress the activation of the opioid receptor more effectively.
To do so, they screened a library of 4.5 billion molecules in the lab in search of molecules that bound to the opioid receptor with naloxone already tucked into the receptor’s pocket. Compounds representing several molecular families passed the initial screen, with one of the most promising dubbed compound 368. Further experiments in cells revealed that, in the presence of compound 368, naloxone was 7.6 times more effective at inhibiting the activation of the opioid receptor, partly because naloxone stayed in the binding pocket at least 10 times longer.
“The compound itself doesn’t bind well without naloxone,” said Evan O’Brien, PhD, the lead author on the study and a postdoctoral scholar in Kobilka’s lab at Stanford. “We think naloxone has to bind first, and then compound 368 is able to come in and cap it in place.”
Even better, compound 368 improved naloxone’s ability to counteract opioid overdoses in mice and enabled naloxone to reverse the effects of fentanyl and morphine at 1/10th the usual doses.
However, people who overdose on opioids and are revived with naloxone can experience withdrawal symptoms such as pain, chills, vomiting and irritability. In this study, while the addition of compound 368 boosted naloxone’s potency, it did not worsen the mice’s withdrawal symptoms.
“We have a long way to go, but these results are really exciting,” McLaughlin said. “Opioid withdrawal likely won’t kill you, but they’re so severe that users often resume taking opioids within a day or two to stop the symptoms. The idea that we can rescue patients from overdose with reduced withdrawal might just help a lot of people.”
Compound 368 is just one of several molecules that show potential as NAMs of the opioid receptor. The researchers have filed a patent on the NAMs, and are working on narrowing down and characterizing the most promising candidates. Majumdar estimates that it will be 10 to 15 years before a naloxone-enhancing NAM is brought to market.
“Developing a new drug is a very long process, and in the meantime new synthetic opioids are just going to keep on coming and getting more and more potent, which means more and more deadly,” Majumdar said. “Our hope is that by developing a NAM, we can preserve naloxone’s power to serve as an antidote, no matter what kind of opioids emerge in the future.”