Zebrafish use surprising strategy to regrow spinal cord
Detailed blueprint of nerve cells’ dramatic changes could help identify ways to heal spinal cord damage
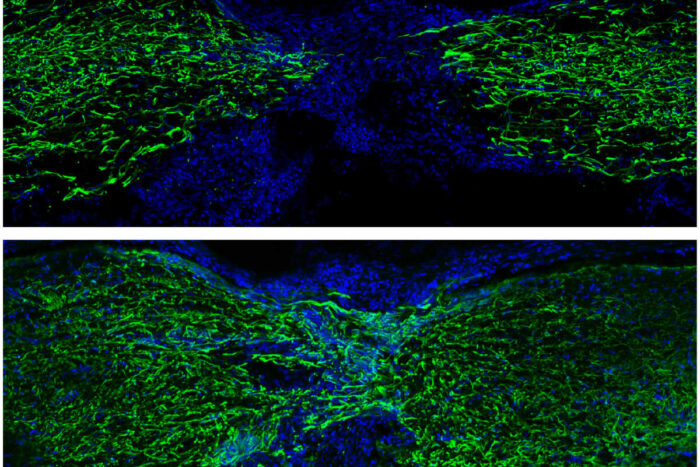 MOKALLED LAB
MOKALLED LABThe top image shows fluorescently labeled cells in the spinal cord of a zebrafish recovering one week after an injury, and the bottom image shows recovery four weeks after an injury. Researchers at Washington University School of Medicine in St. Louis describe the dramatic changes within nerve cells that make regeneration possible. Such findings could inspire the development of new therapies for spinal cord injuries in people.
Zebrafish are members of a rarefied group of vertebrates capable of fully healing a severed spinal cord. A clear understanding of how this regeneration takes place could provide clues toward strategies for healing spinal cord injuries in people. Such injuries can be devastating, causing permanent loss of sensation and movement.
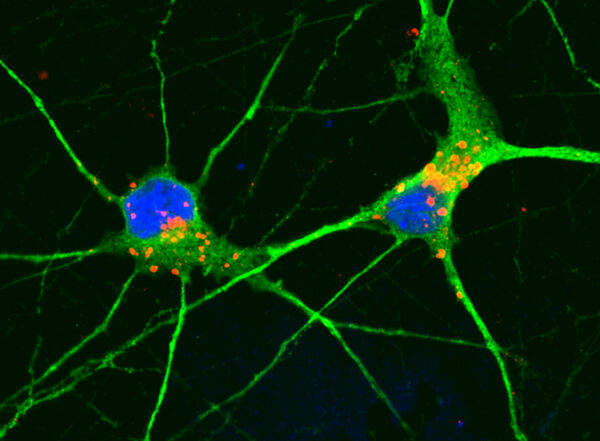
A new study from Washington University School of Medicine in St. Louis maps out a detailed atlas of all the cells involved — and how they work together — in regenerating the zebrafish spinal cord. In an unexpected finding, the researchers showed that survival and adaptability of the severed neurons themselves is required for full spinal cord regeneration. Surprisingly, the study showed that stem cells capable of forming new neurons — and typically thought of as central to regeneration — play a complementary role but don’t lead the process.
The study is published Thursday, Aug. 15, in the journal Nature Communications.
Unlike humans’ and other mammals’ spinal cord injuries, in which damaged neurons always die, the damaged neurons of zebrafish dramatically alter their cellular functions in response to injury, first to survive and then to take on new and central roles in orchestrating the precise events that govern healing, the researchers found. Scientists knew that zebrafish neurons survive spinal cord injury, and this new study reveals how they do it.
“We found that most, if not all, aspects of neural repair that we’re trying to achieve in people occur naturally in zebrafish,” said senior author Mayssa Mokalled, PhD, an associate professor of developmental biology. “The surprising observation we made is that there are strong neuronal protection and repair mechanisms happening right after injury. We think these protective mechanisms allow neurons to survive the injury and then adopt a kind of spontaneous plasticity — or flexibility in their functions — that gives the fish time to regenerate new neurons to achieve full recovery. Our study has identified genetic targets that will help us promote this type of plasticity in the cells of people and other mammals.”
By mapping out the evolving roles of various cell types involved in regeneration, Mokalled and her colleagues found that the flexibility of the surviving injured neurons and their capacity to immediately reprogram after injury lead the chain of events that are required for spinal cord regeneration. If these injury-surviving neurons are disabled, zebrafish do not regain their normal swim capacity, even though regenerative stem cells remain present.
When the long wiring of the spinal cord is crushed or severed in people and other mammals, it sets off a chain of toxicity events that kills the neurons and makes the spinal cord environment hostile against repair mechanisms. This neuronal toxicity could provide some explanation for the failure of attempts to harness stem cells to treat spinal cord injuries in people. Rather than focus on regeneration with stem cells, the new study suggests that any successful method to heal spinal cord injuries in people must start with saving the injured neurons from death.
“Neurons by themselves, without connections to other cells, do not survive,” Mokalled said. “In zebrafish, we think severed neurons can overcome the stress of injury because their flexibility helps them establish new local connections immediately after injury. Our research suggests this is a temporary mechanism that buys time, protecting neurons from death and allowing the system to preserve neuronal circuitry while building and regenerating the main spinal cord.”
There is some evidence that this capacity is present but dormant in mammalian neurons, so this may be a route to new therapies, according to the researchers.
“We are hopeful that identifying the genes that orchestrate this protective process in zebrafish — versions of which also are present in the human genome — will help us find ways to protect neurons in people from the waves of cell death that we see following spinal cord injuries,” she said.
While this study is focused on neurons, Mokalled said spinal cord regeneration is extremely complex, and future work for her team will delve into a new cell atlas to understand the contributions of other cell types to spinal cord regeneration, including non-neuronal cells, called glia, in the central nervous system as well as cells of the immune system and vasculature. They also have ongoing studies comparing the findings in zebrafish to what is happening in mammalian cells, including mouse and human nerve tissue.